Global pandemic demand for overseas nucleic acid testing explodes
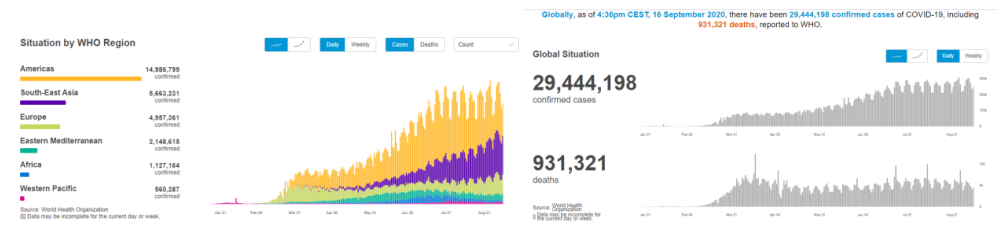
According to WHO statistics, as of 4 pm on September 16, 2020, Beijing time, the number of confirmed cases of COVID-19 worldwide has exceeded 29.44 million and more than 930,000 have died.
Facing the increasingly serious overseas epidemic, the demand for COVID-19 nucleic acid detection reagents is huge. Chinese diagnostic reagent companies have rich clinical experience in product application, and at the same time, they also have a large advantage in cost, providing a huge market opportunity for internationalization. However, the process of exporting kits overseas is complicated and faces many problems.
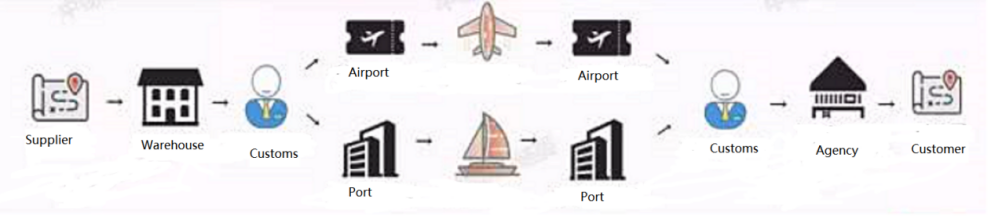
The long-distance cold chain logistics and transportation problem may become the biggest obstacle to export overseas
With the improvement of the country's export policies for anti-epidemic products, and the upgrading of the flow of people and logistics in various countries, the transportation time of reagents has been prolonged and there is uncertainty, and the product problems caused by the transportation of reagents have become prominent. To ensure that the temperature is up to standard and the product quality is qualified, a box of nucleic acid detection reagents of less than 50g and a few kilograms of dry ice can only last for two or three days. The problem of remote cold chain logistics and transportation may become the biggest obstacle to export overseas.
Low temperature requirements result in extremely high transportation costs. Conventional nucleic acid detection reagents need to be stored and transported cold chain at (-20±5)°C to ensure that their biologically active ingredients do not become invalid. In terms of industry practice, the actual weight of the reagents issued by the manufacturer is less than 10% of the box (or far below this value), and most of the weight comes from dry ice, ice packs, and foam boxes, and the transportation cost is extremely high.
Logistics is extended and cold chain effects are discounted. In special periods, the overall transportation of reagent kits has greatly extended the transit time. In order to ensure the quality of conventional liquid reagents, exporters often need to prepare cold chain configurations that are several times the usual domestic transportation. If the transportation temperature cannot be guaranteed, the quality of the reagent products delivered to the customer will be a big question mark.
Insufficient hardware configuration and insufficient storage space. Under normal circumstances, medical institutions routinely carry out molecular testing projects and will not equip too many refrigerators or add large-area cold storage. During the epidemic, there are not many warehouses of charity organizations that can reach -20°C storage conditio
Full-component lyophilized reagents, export nucleic acid reagents to realize normal temperature transportation
In order to break through the bottleneck that molecular diagnostic reagents need to be stored and transported at -20°C, the "Novel coronavirus 2019-nCoV RT-PCR nucleic acid detection kit (lyophilized)" developed by Shanghai Chuangkun Biotech Inc. is a full-component frozen Dry reagents have strong thermal stability to withstand high temperatures of 47°C for at least 60 days, and can be stored at room temperature and transported at room temperature. This effectively solves the pain points that the transportation of liquid COVID-19 nucleic acid reagents requires full cold chain protection in the past, and relieves the pressure on epidemic prevention and control.
Advantages of lyophilized nucleic acid reagents
Shanghai Chuangkun Biotech Inc.'s full-component lyophilized COVID-19
nucleic acid detection reagent has the following advantages in addition
to its composition and activity compared to liquid reagents:
Storage and transportation in the room temperature:it does not need to be stored at a low temperature before opening, which is convenient for medical institutions at all levels.
complete in one step:All components are lyophilized, no PCR reaction system preparation is required, and can be used after reconstitution, greatly simplifying the operation process.
Detect 3 targets at once:The target covers the Novel coronavirus ORF1a/b gene and N gene. In order to reduce false negatives, the IC test of the internal reference gene is added to the product, which can effectively monitor the entire experimental process from sampling, extraction to amplification, and avoid false negatives. Missed inspection.
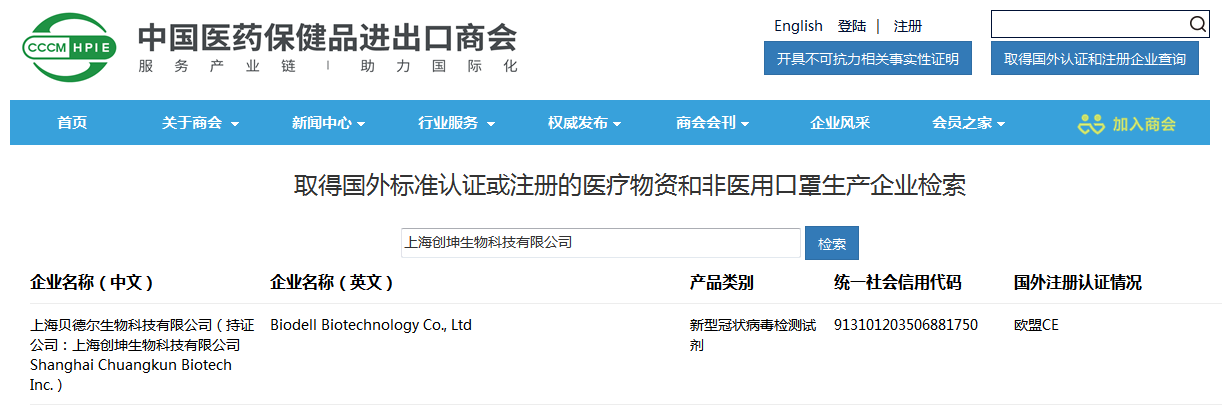
At present, the full-component lyophilized COVID-19 nucleic acid detection reagent has obtained the EU CE certification, and successfully entered the "white list" of the China Chamber of Commerce for Import and Export of Medical Insurance on September 15, 2020, and it means that the kit is officially approved to be exported for overseas sales to help the global fight the COVID-19 epidemic.

Shanghai Chuangkun Biotech Inc.has been listed in the latest "Medical Insurance Chamber of Commerce's List of Medical Material Manufacturers Obtaining Foreign Standard Certification or Registration" issued by the China Chamber of Commerce for Import and Export of Medicines and Health Products. It has obtained export qualifications and can export to support global anti-epidemic.
The virus knows no borders, and the prevention and control of the epidemic requires the joint response of the international community. The application of the full-component freeze-drying process in the COVID-19 nucleic acid detection reagents will be beneficial to the prevention and control of the global COVID-19 epidemic. In order to support the international community to jointly respond to the global public health crisis under the COVID-19 epidemic,Shanghai Chuangkun Biotech Inc. provides high-quality "Novel coronavirus 2019-nCoV RT-PCR nucleic acid detection kit (lyophilized)" to contribute to the global fight against the epidemic Chinese power!
Post time: Oct-28-2020

 中文
中文